These eye drops were sold at major retail chains across the US, including CVS, Walgreens, and Kroger, ABC News reported on 4/4. The recalled products were manufactured by KC Pharmaceuticals Inc., based in Pomona, California, and marketed under several popular brands such as Best Choice, CVS, Discount Drug Mart, Gericare, and Walgreens.
The recall began on 3/3, but the FDA officially classified the incident as Class II on 31/3. According to the FDA's definition, Class II recalls involve products that may cause temporary or reversible health problems, with a low probability of serious adverse health consequences.
In total, over 3.1 million bottles across 8 different product lines are affected. These include redness relief eye drops, lubricating dry eye drops, soothing formulas, and artificial tears, each in 15 ml bottles. A full list, including brand names, lot numbers, UPCs, and expiration dates, is available on the FDA's official website.
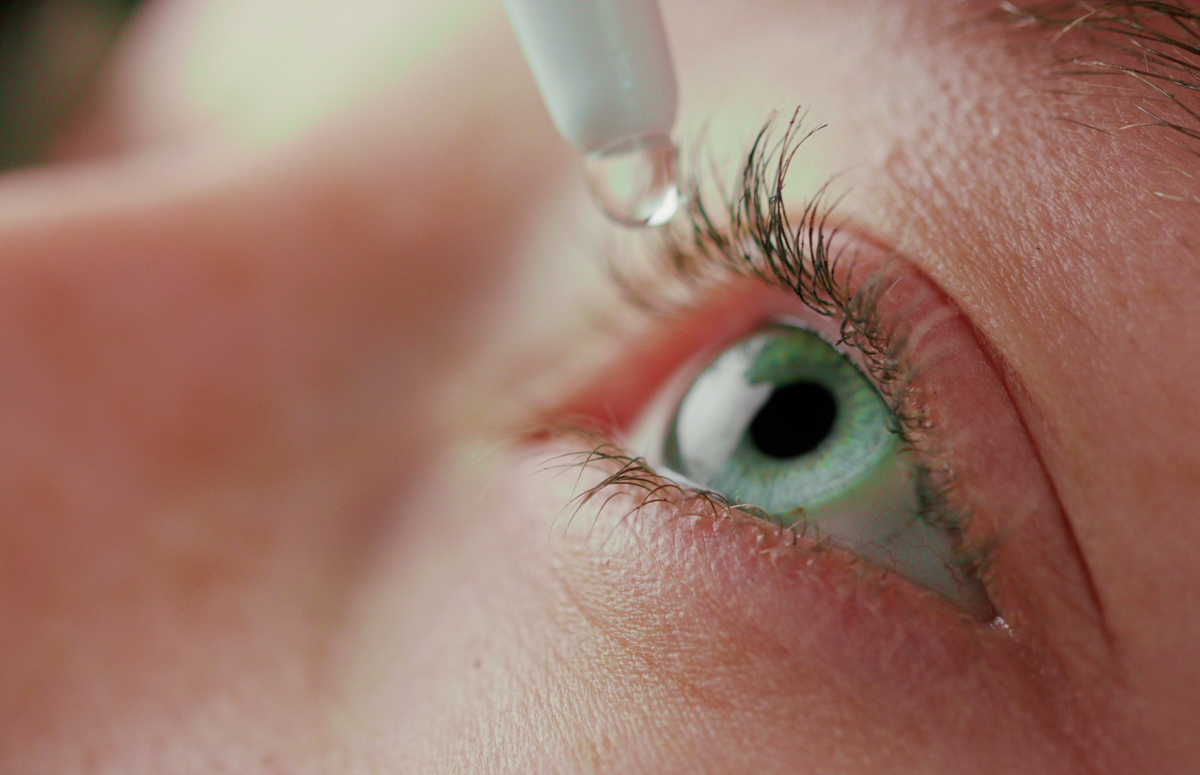 |
Illustration: Mputsylo - stock |
CVS announced the recall of several product lines on its website, including GeriCare artificial tears and CVS-branded redness relief and dry eye drops (including two-pack formulations). The stated reason is the risk of microbial contamination.
In a statement sent to ABC News on 3/4, a CVS spokesperson stated that 4 of these product lines "ceased business nearly a year ago", and the company is "working closely" with the manufacturer to implement the recall.
"We are committed to providing products that are safe, perform as intended, comply with health regulations, and meet our customers' needs", the representative affirmed.
CVS also stated that customers who purchased the affected products can return them to any CVS pharmacy for a full refund.
Binh Minh (According to ABC News, NBC News)