The affected batch bears registration number 893100060724 and code 10370725. It was manufactured on 14/7/2025, with an expiry date of 14/1/2028. The exact quantity subject to recall is currently unknown. Natri clorid 0,9% is a common physiological saline solution used for eyes and nose, widely available in pharmacies across the country.
The regulatory agency has instructed Pharmedic to immediately halt sales, quarantine all affected products, and complete the recall process within 30 days. The company must bear all costs associated with the handling of the recalled products and report the recall results to the health sector.
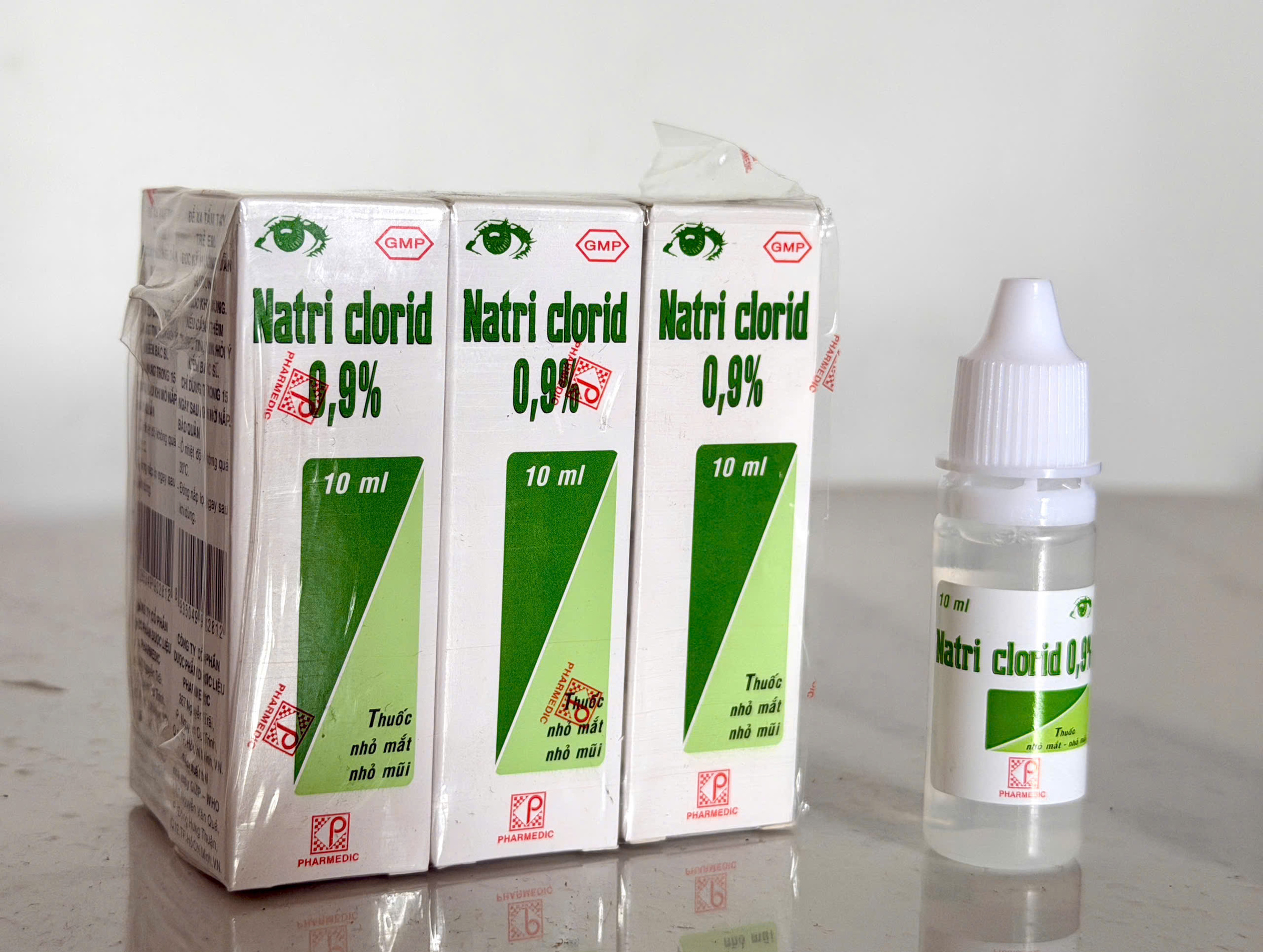 |
A batch of Natri clorid 0,9% eye drops produced by Pharmedic. Photo: *Le Nga*. |
This recall follows evaluations conducted by the Quang Ngai Province Center for Drug, Cosmetic, and Food Testing and the Ho Chi Minh City Institute of Drug Testing. These units collected market samples and discovered the solution was not transparent, containing visible sediment, suspended particles, or other impurities.
Authorities have classified this as a quality violation of 'muc do ba'. While the health sector considers this a low-risk violation, posing no immediate threat to life or severe consequences, the sensitive nature of the eyes means that using an impure solution can easily cause irritation, damage, or reduce the effectiveness of treatment.
The Drug Administration of Vietnam advises individuals, pharmacies, and medical facilities to immediately cease distributing and using the affected saline solution batch. They are also urged to promptly return the products to their respective suppliers.