Regulatory authorities issued the fine decision on 21/4. In addition to paying the penalty, the Hanoi-based company must withdraw the declaration receipt numbers for all non-compliant product batches. The list of products slated for destruction includes sunscreens, facial cleansers, skin creams, and body lotions from well-known brands such as Anessa, Hatomugi, Rosette, Skin Aqua, Senka, Shiseido, Hada Labo, and Aqua Label.
Regulatory agencies imposed stricter penalties because the company violated regulations across multiple product categories simultaneously. During the inspection, the company failed to present its Product Information File (PIF). While current law does not mandate submitting a PIF during product declaration for circulation, distributors are required to retain and provide this document upon request by regulatory bodies to demonstrate product quality, safety, and efficacy.
The Drug Administration of Vietnam (DAV) instructed the company, legally represented by Le Viet Nam, to pay the fine within 10 days. The company has 30 days to complete the recall and destruction of the products and submit a report on the results. Non-compliance will lead to enforcement measures by authorities. Furthermore, the DAV suspended the processing of new cosmetic product declarations from the company for 6 months.
In recent times, regulatory agencies have consistently ordered businesses to withdraw hundreds of cosmetic products from the market due to inaccurate documentation, substandard quality, or the presence of prohibited ingredients. Ta Manh Hung, Deputy Director of the Drug Administration of Vietnam, stated that the health sector is intensifying its oversight of market products. This initiative aims to reduce the percentage of non-compliant cosmetics to below 1.5% among over 3,000 samples inspected by authorities.
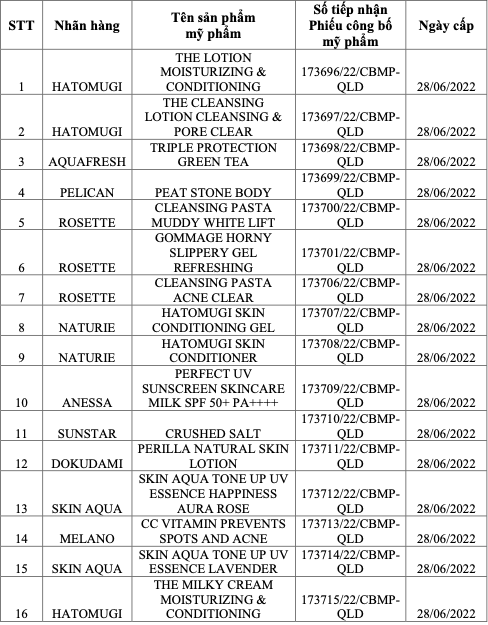 |
List of recalled cosmetic products. Photo: Drug Administration of Vietnam |
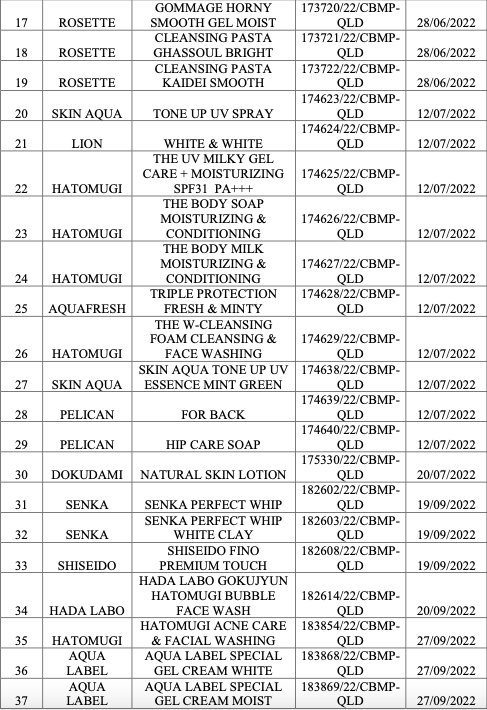 |
List of recalled cosmetic products. Photo: Drug Administration of Vietnam
Le Nga