The Envacgen (EV71) vaccine, manufactured in Taiwan, is expected to be deployed later this year and will be used for children aged two months to under six years. Previously, a phase three clinical trial involving over 3,000 children, 80% of whom were Vietnamese, demonstrated a protective efficacy of 96,8% against the EV71 strain. Long-term follow-up data also confirm that the immune response is maintained for at least five years and continues to be monitored.
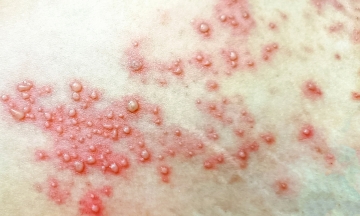
Hand, foot, and mouth disease circulates year-round, typically increasing during spring and summer, primarily affecting children under five years old. In 2025, Vietnam recorded over 107,000 cases, an increase of nearly 29% compared to the previous year. Entering 2026, many localities continue to experience outbreaks, with TP HCM alone reporting over 6,400 cases.
 |
Illustration of EV71 vaccine use. Photo: Vecteezy |
EV71 is the most dangerous strain, linked to most severe cases and deaths due to neurological and respiratory complications. Major outbreaks in 2011, 2021, and 2023 all showed strong circulation of EV71. In 2023 alone, there were over 180,000 cases and 31 deaths. Currently, there is no specific treatment.
According to the World Health Organization (WHO), the EV71 vaccine does not protect against all strains of the hand, foot, and mouth disease virus. Therefore, in addition to vaccination, families still need to implement and teach children other preventive measures such as hand washing, sanitizing toys, disinfecting surfaces, and avoiding contact with sick individuals. If a child shows signs of fever or blisters on their hands, feet, or mouth, they should be taken for examination immediately.
Van Ha