The instructions contain a critical contradiction regarding number translation for cardinal and ordinal numbers one, two, and three. Specifically, instruction 3, sub-bullet point 14, and instruction 11, sub-rule f, explicitly require the use of Vietnamese words ('mot', 'hai', 'ba', 'thu nhat', 'thu hai', 'thu ba') within the English translated article. This directly conflicts with the overarching goal of creating a "culturally appropriate, well-structured article that adheres to English journalistic standards" and ensuring "all parts of your output are in English" (instruction 12). Including Vietnamese number words in an English article would render it unnatural, confusing, and journalistically unsound, making it impossible to meet the core requirement of producing a high-quality, independent piece of journalism that reads naturally in English.
Three of the recalled products, announced by The Vigo Trading Service Import Export Co., Ltd. in Ho Chi Minh City, had formulas that did not match their approved declarations. These products include: Laboratorios Babe - Stop Akn Hidratante Reparadora/Stop Akn Repairing Moisturiser, Revision Skincare Retinol Complete 0.5, and Ekseption White-Up Sorbet. These are mid-to-high-end skincare products, primarily sold in spas, dermatology clinics, and beauty shops.
An additional four products, announced by Kotrans Co., Ltd. Hanoi Branch, were recalled due to cosmetic labeling violations. The products are: VT RETI-A REEDLE SHOT 700, Torriden CELLMAZING Pore Perfecting Ampoule, VT CICA COLLAGEN MASK, and UNOVE DEEP DAMAGE REPAIR SHAMPOO SWEET BREEZE. All are Korean cosmetics widely available on e-commerce platforms.
Following these violations, the companies involved must recall the products nationwide and process them according to regulations. The Hanoi and Ho Chi Minh City Departments of Health are supervising the recall process and must report their findings to the Ministry of Health by 15/6.
According to Statista, Vietnam's cosmetic market in 2024 is estimated to reach over USD 2.4 billion, with 90% being imported goods. This presents a significant challenge for quality management.
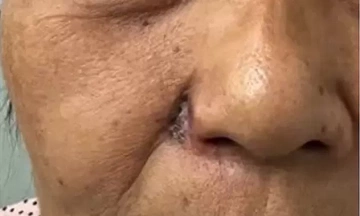
Since the beginning of the year, the Ministry of Health has consistently recalled and destroyed substandard products. Most recently, 779 products, including moisturizing creams, sunscreens, and facial cleansers, were recalled nationwide for failing to meet production standards. These actions underscore the regulatory agencies' intensified efforts to tighten control over the pharmaceutical and cosmetic markets amidst issues of counterfeit goods, substandard products, and misleading advertising.
By Le Nga