**Answer:**
Children should not receive vaccines while suffering from hand, foot, and mouth disease because their bodies are experiencing an acute viral infection, requiring priority for treatment and monitoring. Additionally, after vaccination, children may experience mild reactions such as pain, swelling, and redness at the injection site, slight fever, and fatigue. These reactions are typically mild and resolve spontaneously within one to two days. Vaccinating a child who is already ill can cause these post-vaccination reactions to overlap with existing symptoms, making monitoring difficult and potentially reducing the vaccine's protective efficacy.
Children must postpone vaccination and isolate for at least 10 days, returning to normal activities only after their fever has subsided, mouth sores have healed, blisters have dried, and there are no neurological or cardiovascular complications. Vaccination should only occur when the child has fully recovered and is in good health.
Furthermore, bringing an ill child to a vaccination center risks transmitting the virus to others. The virus is present in nasal and throat secretions, saliva, and feces, making it easily spread through coughing, sneezing, direct contact, or by adhering to surfaces of objects.
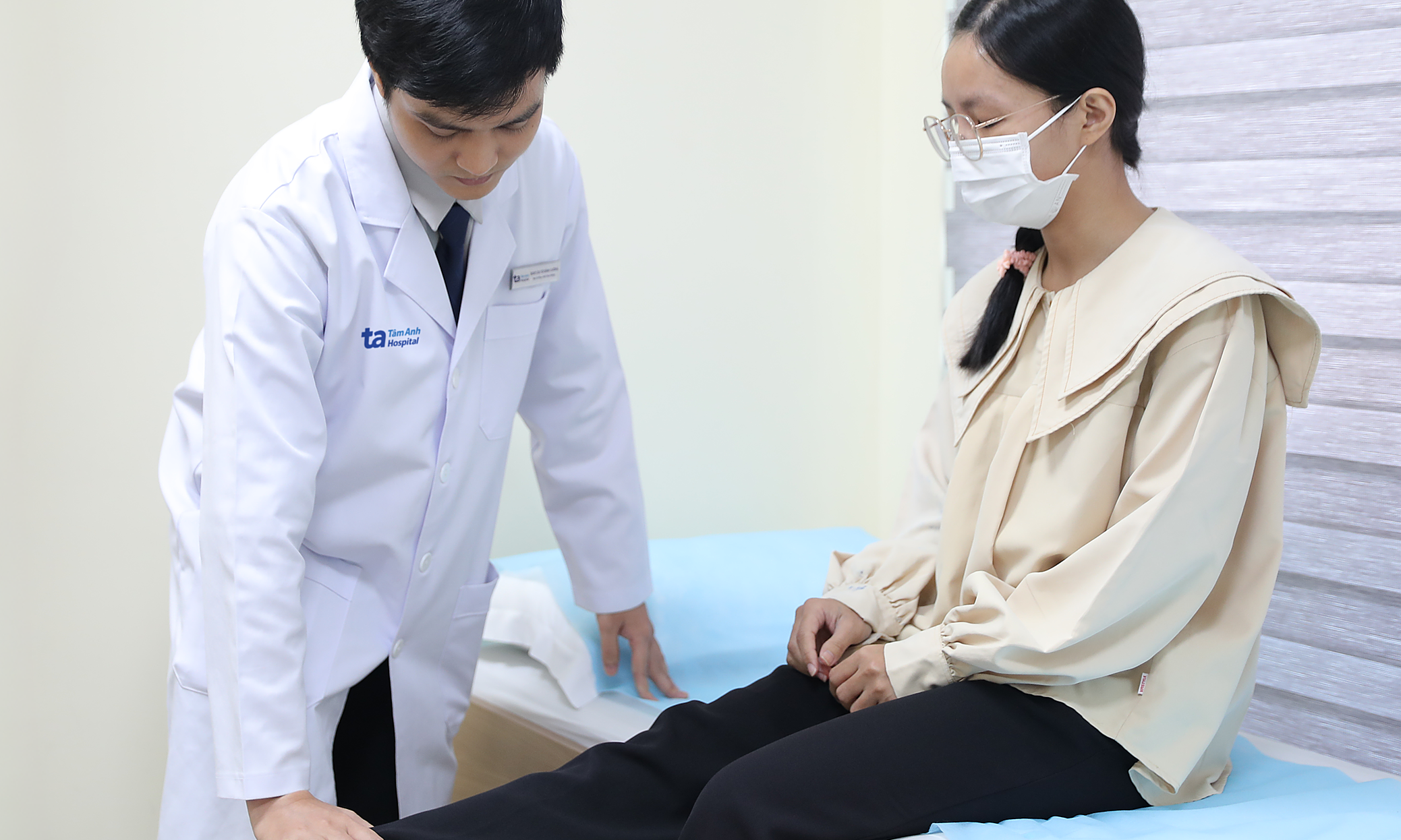
During the illness, children with mild cases can be cared for at home. Parents should monitor their fever, diet, alertness, maintain hygiene, and ensure adequate hydration. If a child experiences persistent high fever, frequent vomiting, startling, lethargy, cold hands and feet, unsteady gait, or unusual irritability, immediate medical attention is necessary, as these could indicate neurological, cardiovascular, or respiratory complications.
 |
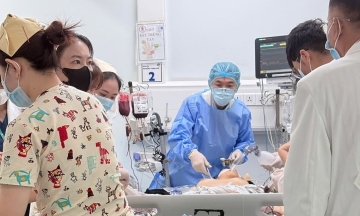
Children receiving vaccines at the VNVC Immunization System. Photo: Binh An |
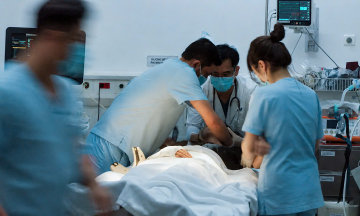
Hand, foot, and mouth disease is an acute viral illness transmitted via the digestive tract and direct contact. It commonly affects children under five years old and spreads rapidly in schools and daycare centers. Most cases are mild, presenting with fever, mouth sores, and a blistering rash. However, some cases can quickly become severe, especially when caused by the EV71 strain, which can lead to dangerous complications.
In 3/2026, the Ministry of Health approved an EV71 vaccine for children aged two months to under six years old. This vaccine demonstrates high efficacy and cross-protection against circulating genotypes in Vietnam. The VNVC Immunization System is currently implementing plans to make this vaccine available in Vietnam soon.
Beyond hand, foot, and mouth disease, parents should also proactively prevent respiratory or contact-transmitted illnesses such as meningococcal disease, chickenpox, influenza, pertussis, measles, and others. Vietnam currently has a comprehensive range of vaccines available to help children reduce the risk of contracting multiple infectious diseases simultaneously.
Master of Science, Doctor Nguyen Van Quang
Medical Manager, VNVC Immunization System
Readers can submit vaccine consultation questions for doctors to answer here.