On 14/4, the Drug Administration of Vietnam determined that Lotus An Viet Co., Ltd., based in Hai Phong, had violated cosmetic management regulations. The company failed to present a Product Information File (PIF) during an inspection. This file is a mandatory technical document businesses must retain to verify product quality, safety, and effectiveness.
Regulations do not require businesses to submit a PIF when initially declaring a product for circulation. However, the company responsible for marketing the product must present this file immediately upon request by authorities. Failure to provide the PIF results in the revocation of the product's registration number and subsequent penalties.
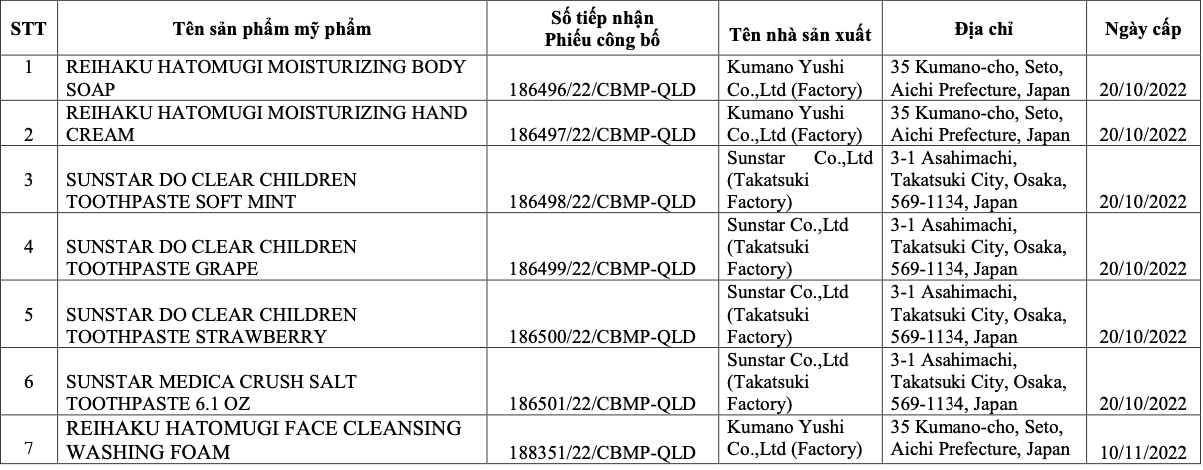 |
List of seven recalled cosmetic products. Photo: Drug Administration of Vietnam provided |
Consequently, the company received a fine of 75 million dong. This penalty was intensified because seven products were found in violation simultaneously during the inspection. Beyond the monetary fine, the business must revoke seven cosmetic product announcement registration numbers and recall and destroy all seven non-compliant products, which include: shower gels, hand creams, toothpastes, and facial cleansers.
Lotus An Viet Co., Ltd. is responsible for notifying distributors of the recall, accepting returned products, and ensuring their destruction. The company must submit a report on the recall results to the Drug Administration of Vietnam before 9/5. The Hai Phong Department of Health will oversee the company's recall and destruction activities.
Recently, regulatory agencies have ordered the recall of hundreds of cosmetic products due to issues such as incorrect declaration files, substandard quality, or the presence of unapproved ingredients. Ta Manh Hung, Deputy Director of the Drug Administration of Vietnam, noted that these recent recalls indicate a more stringent approach by regulatory bodies to control the quality of products on the market. This increased oversight has led to a reduction in the rate of quality-violating cosmetic products being recalled, now below 1.5% among over 3,000 market samples inspected.
The agency also advises consumers to purchase pharmaceuticals and cosmetics only from legal businesses with verified origins. It recommends checking product information on the agency's official website before use.
Le Nga